Ilmenite is converted into pigment grade titanium dioxide via either the sulfate process or the chloride process. Both Sulfate and Chloride Processes produce the titanium dioxide pigment in the rutile crystal form, but the Sulfate Process can be adjusted to produce the anatase form.
/ MANUFACTURE OF TITANIUM DIOXIDE.PROCESSING TITANIUM DIOXIDE.Titanium dioxide has remarkable optical properties, with a very high refractive index close to that of diamond. It is also a very stable compound and can be heated to over 2000 K before melting. It is these properties that underlie its most important uses.Uses of titanium dioxideTitanium dioxide is the most used white pigment and provides whiteness and opacity for paints and coatings (including glazes), plastics and paper.It has many speciality uses. It is resistant to UV radiation and thus does not discolour over a long period of time and ultra pure and fine crystal titanium dioxide grades are being increasingly used for sun screens.It is also being used in novel nanotechnology applications.Figure 1 Uses of titanium dioxideThe proportions shown in Figure 1 are for the World. The figures vary country by country.
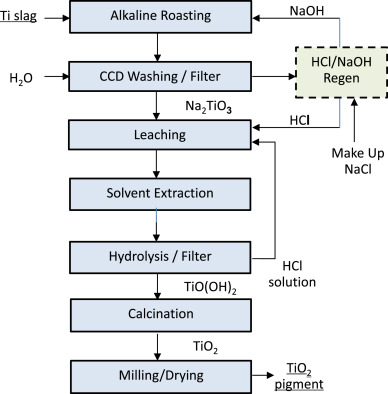
For example in the US, the corresponding figures for the uses of titanium dioxide in paint is 50%, whereas those for plastics and paper are similar, 25% and 13%.Much smaller amounts of titanium dioxide are used as a semi-conductor and to catalyse the photodecomposition of water into hydrogen and oxygen.It also has a high dielectric constant and a high resistance and thus it is used to make capacitors.The strong bonding between titanium and oxygen gives great thermal stability, melting at 2100 K. It is thus used as a ceramic material. It is also used to increase the acid resistance of vitreous enamels. Annual production of titanium dioxide World5.1 million tonnesEurope1.4 million tonnesUS1.4 million tonnesChina1.7 million tonnesManufacture of titanium dioxideThere are two main processes, the Sulfate Process and the Chloride Process (Table 1) which use the two principal ores, ilmenite and rutile, respectively. Ilmenite contains 45-60% TiO 2 and rutile contains up to 99% TiO 2. The ores are mined worldwide but most production is in Australia and South Africa.Each large producer of titanium dioxide balances its production between the two processes. Each produces the oxide in the rutile crystal form but the Sulfate Process can also produce another form of the oxide, anatase, which is softer and which is used for a small number of specialist applications.The Sulfate Process is run as a batch process; the Chloride Process is run as a continuous process.It is estimated that about 65% of the world's production is based on the Chloride Process.
Sulfate ProcessChloride Processlong established and simple technologynew technologyuses lower grade, cheaper oresneeds high grade orebatch processcontinuous processlarge amounts of waste materialssmall amounts of waste formed with toxicity problems: Cl 2 and TiCl 4pollution control expensiverecovery and recycling of chlorine possibleproduces anatase and rutile pigmentsonly produces rutile pigmentsTable 1 Comparison of the two processes for the manufacture of titanium dioxide. The Sulfate ProcessThe chemistry of the process involves three main stages:a) dissolving the oreb) formation of hydrated titanium dioxidec) formation of anhydrous titanium dioxide (a) Dissolving the oreThe ore is usually ilmenite, FeTiO 3. It is ground finely and dissolved in sulfuric acid to form a mixture of sulfates:Before the titanium dioxide is extracted, the iron ions must be removed from the solution so that the colour of the final product is not spoiled. The solution is therefore reacted with recycled iron sources to convert any iron(lll) ions that may be present to iron(ll) ions:The solution is allowed to stand so that the unreacted solid settles, and the clear liquid is poured off before being concentrated by evaporation.
Cooling then allows light green crystals of iron(ll) sulfate to form and to be filtered off. These are sold separately. The remaining solution contains titanyl sulfate, TiOSO 4.(b) Formation of hydrated titanium dioxideThe next stage involves the hydrolysis of the titanyl sulfate in solution to give insoluble, hydrated titanium dioxide:This is a critical stage and the conditions must be controlled to ensure that the precipitate is suitable for filtering and roasting. (c) Formation of anhydrous titanium dioxideThe final stage of the process is the heating of the solid in a furnace, known as a calciner. This is a rotating cylinder which is typically heated by gas flames. As the cylinder turns, the titanium dioxide passes along it and its temperature rises from 313 K, as it enters, to over 1000 K as it leaves:Heating evaporates the water and decomposes any remaining sulfuric acid in the solid. After cooling, the product is 'milled' to form crystals of the size needed.
Crystals may also be coated with another substance, such as aluminium oxide or silica, to make the titanium dioxide mix more easily with liquids or to make the water-based paints made from it last longer.This coating is achieved by dispersing the dry product from the calciner in water containing the dissolved coating chemicals which precipitate from solution onto the TiO 2 crystals. The coating is usually between 3 and 8% by weight in the final dried pigment. This coating is achieved by changing the temperature and pH of the solution. Each TiO 2 crystal needs to be coated uniformly to maximize the effectiveness of the coating. The coated TiO 2 crystals are filtered from the water and dried before being packed for dispatch to the final customer. The Chloride ProcessThere are two main stages:a) the conversion of rutile to titanium(IV) chlorideb) the oxidation of titanium(IV) chloride (a) The conversion of rutile to titanium(IV) chlorideThe rutile is fed into a heated bed together with a source of carbon, usually coke. Chlorine is fed into the bed and the reaction takes place to form titanium(IV) chloride in the vapour form which is removed from the bed.
Iron and other metals in the ore are chlorinated and also leave the bed in the vapour state. The oxygen in the ores is combined with the carbon to form carbon monoxide and dioxide.
The vapour stream is cooled and the metal chlorides other than titanium(IV) chloride are condensed and solidified. The titanium(IV) chloride vapour, which contains almost pure titanium(IV) chloride and has a lower boiling point, is then condensed and stored as liquid. It is then reboiled and distilled to give a purer product to feed to the next stage. (b) The oxidation of titanium(IV) chlorideLiquid titanium(IV) chloride is vaporised and burnt in oxygen, together with a hydrocarbon fuel source (for example, methane) to a high temperature to initiate the reaction and keep the temperature high enough for the reaction to proceed:The titanium dioxide is formed (by adding seed crystals) as a fine solid in the gas stream and is filtered out of the waste gases using cyclones or filters. Once again control of crystal growth is important to give particles of the correct size for pigments. This is done by adding nucleating agents to the gas stream (e.g.
Water or aluminium chloride) and by cooling the products. The chlorine in titanium(IV) chloride is released and recycled to the chlorination stage of the process above.The product contains small amounts of absorbed chlorine gas which are removed. The product is washed and dried before milling and surface treatment in an identical manner to that used in the Sulfate Process described.RELATED TAGS: WHAT IS TITANIUM DIOXIDE,WHERE TO USE TITANIUM DIOXIDE,MAKING TITANIUM DIOXIDE,USES OF TITAN DIOXIDE,TITAN DIOXIDE TO MAKE,TITANIUM DIOXIDE TO MANUFACTURE,TITANIUM DIOXIDE PRODUCTION,USING TITAN DIOXIDE,HOW TO COMPOUND TITANIUM DIOXIDE,PROPERTIES OF TITAN DIOXIDE,TYPES OF TITANIUM DIOXIDE,TITANIUM DIOXIDE SPESIFICATION.SOLVER CHEM.
Titanium dioxide is the whitest pigment widely used in the manufacture of paints and enamels. It may be manufactured by two major processes: the chloride and the sulfate process.These processes were compared and the chloride process was selected and analyzed.
Titanium dioxide containing feed stock is subjected to high temperature carbo chlorination to produce titanium tetra chloride, which is thereafter converted to titanium dioxide by oxidation.This project involves the studying and collecting all preliminary information required for Titanium dioxide manufacture, generating mass and energy balances for the plant, designing the various equipment used in the plant and estimating the cost involved in setting up the plant.Titanium dioxide is produced either in the anatase or rutile crystal form. Most titanium dioxide in the anatase form is produced as a white powder, whereas various rutile grades are often off-white and can even exhibit a slight color, depending on the physical form, which affects light reflectance. Titanium dioxide may be coated with small amounts of alumina and silica to improve technological properties.Commercial titanium dioxide pigment is produced by either the sulfate process or the chloride process. The principal raw materials for manufacturing titanium dioxide include ilmenite.
(FeO/TiO2), naturally occurring rutile, or titanium slag. Both anatase and rutile forms of titanium dioxide can be produced by the sulfate process, whereas the chloride process yields the rutile form. Titanium dioxide can be prepared at a high level of purity. Specifications for food use currently contain a minimum purity assay of 99.0%. Titanium dioxide is the most widely used white pigment in products such as paints, coatings, plastics, paper, inks, fibers, and food and cosmetics because of its brightness and high refractive index ( 2.4), which determines the degree of opacity that a material confers to the host matrix.
When combined with other colors, soft pastel shades can be achieved. The high refractive index, surpassed by few other materials, allows titanium dioxide to be used at relatively low levels to achieve its technical effect. The food applications of titanium dioxide are broad. US regulations authorize its use as a color additive in foods in general at levels not to exceed 1%. The European Union also permits its use in foods, in general, with some specified exceptions, at quantum satis levels. India restricts its uses to chewing gum and bubble gum at not more than 1% and to powdered concentrate mixes for fruit beverage drinks not to exceed 100 mg/kg.
Japan lists its use as a food color without limitation, other than specifying certain foods in which it is not permitted. Finally, titanium dioxide is listed in Table 3 of the Codex General Standard for Food Additives, which lists additives that may be used in food, in general, unless otherwise specified, in accordance with GMP.ORES AND NATURAL OCCURRENCETitanium is the world's ninth most abundant element. Titanium dioxide occurrences in nature are never pure, it occurs in complex oxides, usually in combination with iron and also with the alkaline earth elements. These impurities color the ores from buff to black and they are therefore far removed from the requirements of a clean white titanium dioxide pigment. The important ores are. Ilmenite (FeTiO3). Mineral Rutile tetrag (TiO2).
Brookite rhombic (TiO2). Perovskite (CaTiO3).
Sphene (CaTiSiO5)The oxides can be mined and serve as a source for commercial titanium. The original commercial ore, first used by the sulfate process is ilmenite. This occurs mainly as a black sand: in Australia, South Africa, India, the USA and Malaysia, and also as a hard rock deposit: in Canada, Norway and the USA. Ilmenite typically contains 45-65% TiO2 with iron making up the majority of the remainder. The second most available ore is the buff colored mineral rutile, which contains around 95% TiO2 with smaller amounts of iron and other impurities. Rutile sands are mostly found in Australia, Sierra Leone and South Africa. These are produced either by acid leaching of the iron and other impurities or via a slagging process to separate molten iron, producing a Titania rich slag which is then solidified and ground to the desired particle size.
This beneficiation process allows fine ore sand to be converted to a more desirable, coarser TiO2 feed stock with iron produced as a by-product.Titanium dioxide is one of the most important chemicals, finding a variety of applications in the Paint and Paper industries and many other fields. For the manufacture of Titanium dioxide in this project, we have used the Chloride process, a process that has many advantages over its alternative process. As the first step, preliminary analysis was carried out on the basis of properties and applications of Titanium dioxide and the various processes used in its manufacture. The material and energy balances for the various operations involved in the process were done. Next, the equipment's involved in the manufacturing process were designed. Cost estimation was done and the payback period for the operation of the plant was calculated.
The plant layout and locations were decided upon to complete the conception of the whole process of manufacture of Titanium dioxide.Please go through the attachment for complete project implementation.
